When we talk about climate change, it is often that we talk about CO2 emissions. The C in CO2 stands for carbon, an element we that's found everywhere: in our bodies, in nature, and in fuels. Carbon comes in chains and all the connections that carbon makes with other molecules contain energy. The energy content of such a bond depends on which other elements carbon is connected to. A connection with Hydrogen and Carbon, for example, has a high energy content, and if we only connect Carbon to Oxygen – as in CO2 – the energy is minimal.
When we use fuels, what we basically do is to break carbon bonds and replace them with carbon-oxygen bonds. As these carbon-oxygen bonds have less energy, we can use the leftover energy, which usually translates as heat, as we please.
When it comes to the catastrophic environmental impact of our fuel consumption, the above-described process itself is not the problem. There are ways to reverse the process and keep the balance between burning fuel and making fuel. In nature, photosynthesis provides this balance. The problem with our fuel consumption lies in where we are getting the fuel.
At the moment, we mainly source our fuel from the ground where carbon has been stored for millions of years after photosynthesis has turned it into fuel. By digging it out at an unprecedented speed, we are bringing this equilibrium out of balance as nature’s photosynthesis can’t keep up with the amount of fuel we are burning. And ultimately, this equilibrium – which we destroy – determines the climate of our very own planet.
To turn things around and protect the equilibrium, we need to put as much energy into the CO2 as we have taken out of the fuel. In this process, we will have losses; therefore, in the end, we will likely need way more energy than we can store. Plants get this energy from the sun, ideally, we can do the same. In any case, we need to use renewable energy sources such as solar or wind power. This way we can make the fuel cycle circular and also solve the storage problem that comes with renewables. In times where the wind blows and the sun shines, we can produce fuels with a high energy density and use them on cloudy wind-still days.
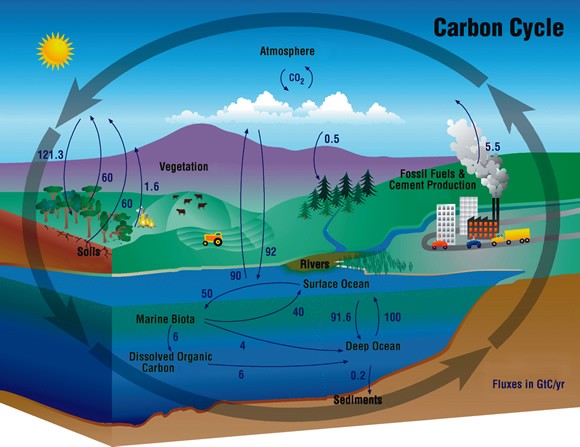
In order to make sure the atmosphere doesn't have a destructive amount of carbon dioxide, we need to convert much more CO2 back into fuels compared to what we burn. It's not hopeless, we do have ways to actually do it. The first option to do so would be using what we already have: photosynthesis. We could make fuels from plants which have grown on converted CO2. As a second option, we can find processes that can turn CO2 back into fuels – just as nature does.
The good news is that these Carbon (based) fuels can already be made on a test scale. Various different routes involving Methane or Formic acid as intermediates are available Usually the CO2 is converted to in multiple chemical steps. The CO2 can already be captured efficiently from industrial plants as the power or cement industry. It is just not done on a bigger scale as the producers have no one to sell their excess CO2 to yet. Additionally, we could even get CO2 directly from the air – just as plants do.
The options above are solutions to make our fuel circular in the future whilst still using chemical fuels in our engines. On top of that, from the processes described above, we can not only obtain fuels but also chemicals that we won’t need to bring back into the cycle and therefore can store the CO2 much longer. Sadly all these projects still need a lot of development and the chemical fuel industry is known to be rather conservative and slow moving. Now, more than ever, we need more awareness and incentives to push the industry to develop and produce green fuels.
This article was written by Eric Schuler for Kinder World. Schuler is a PhD candidate at the University of Amsterdam and works on new industrial sustainable chemistries to turn captured CO2 into useful things such as plastics or fuel. He's also a photojournalist with an interest in climate and land-use change.




